Lack of comprehensive understanding of immune landscape evolution across the human lifespan addressed through integrated multi-omics approach
January 29, 2025 — A groundbreaking study led by Prof. Pu Jun’s team at Shanghai Jiao Tong University, published in Nature Immunology (IF=27.8), has mapped the dynamic evolution of human peripheral immune cells across the entire lifespan—from birth to old age—using an innovative integration of single-cell RNA sequencing (scRNA-seq), T cell/B cell receptor (TCR/BCR) sequencing, and mass cytometry (CyTOF). The research, titled “Integrating single-cell RNA and T cell/B cell receptor sequencing with mass cytometry reveals dynamic trajectories of human peripheral immune cells from birth to old age,” provides unprecedented insights into how the immune system matures, functions, and declines over time.

Key Findings
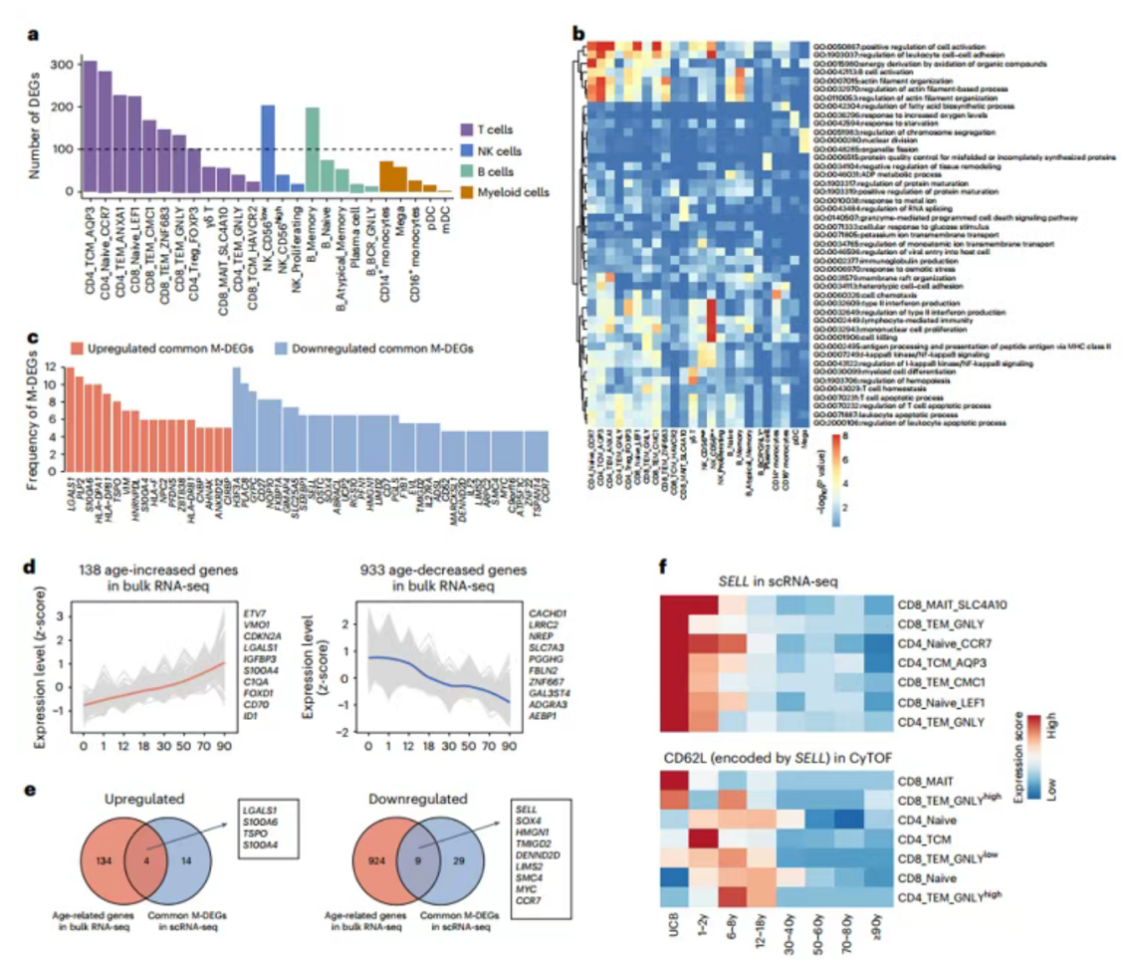
1. T Cells Show Age-Driven Divergence
T cell subsets exhibited the most pronounced age-related changes. Among the top 10 immune cell populations with significant transcriptional shifts, 8 were T cell subtypes (e.g., CD4_Naive_CCR7, CD8_TEM_GNLY). These changes were linked to inflammation, metabolic pathways, and immune regulation.
Clonal Expansion Peaks in Youth and Old Age: Effector memory T cells demonstrated biphasic clonal expansion, peaking in childhood (2–12 years) and late adulthood (70–90 years). Early clones were associated with cytotoxicity, while older clones correlated with chronic inflammation and survival signals.
Naive T Cell Decline: CD4+ naive T cells dropped sharply in childhood and stabilized, whereas CD8+ naive T cells declined continuously from adolescence to old age, accompanied by reduced TCR diversity.
2. MAIT Cells Peak in Adolescence
Mucosal-associated invariant T (MAIT) cells reached functional and numerical peaks during adolescence (18 years), marked by high antimicrobial gene expression (e.g., GNLY, KLRB1) and enhanced pathogen clearance in lab experiments.
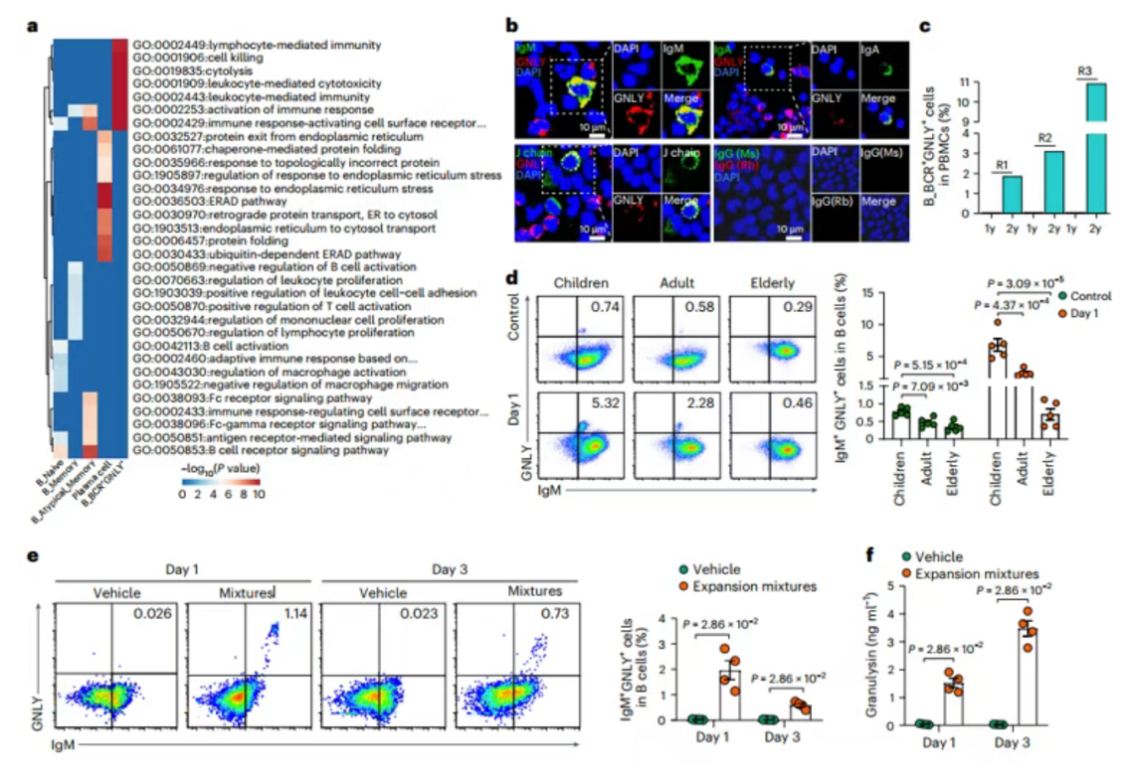
3. Discovery of Cytotoxic B Cells in Children
A novel B cell subtype (B_BCR+GNLY+), co-expressing cytotoxic markers (e.g., GNLY) and B cell receptors, was identified. These cells were enriched in early childhood (2–6 years) and demonstrated antimicrobial activity via granulysin secretion.
4. Immune Age Prediction Model (siAge)
A machine learning model, siAge, accurately predicted immune age using 21 key genes (Pearson r=0.994). It detected significant deviations in autoimmune diseases (e.g., lupus, Kawasaki disease), highlighting clinical potential for early diagnosis.
Bridging Developmental and Aging Immunology
Human immune systems undergo maturation after birth, enhancing pathogen defense, but later face age-driven decline linked to infections, cardiovascular diseases, and cancer. Previous studies using bulk sequencing or limited age ranges lacked resolution to capture lifelong immune dynamics. This study fills critical gaps by integrating multi-omics approaches across 13 age groups, offering a holistic view of immune cell composition, transcriptional and proteomic prohttps://www.renji.com/files, clonal dynamics, and intercellular communication over nine decades.
Implications and Future Directions
“Our work provides a rich resource for understanding how peripheral immune cells evolve through development, maturation, and aging,” said Prof. Pu Jun. “The siAge model could revolutionize how we evaluate immune health and intervene in age-related diseases.” The findings also suggest that aging immune cells may contribute to systemic organ aging, opening new avenues for research into longevity and regenerative medicine.
Journal Reference: Nature Immunology (2025). DOI: 10.1038/s41590-024-02059-6